Leptophrys vorax (Cienkowski, 1865) Zopf, 1885
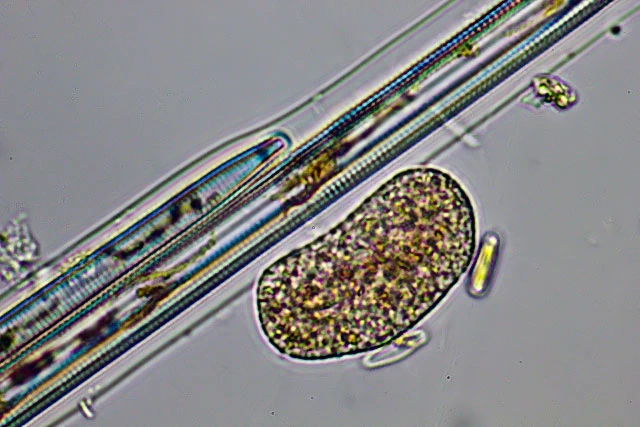
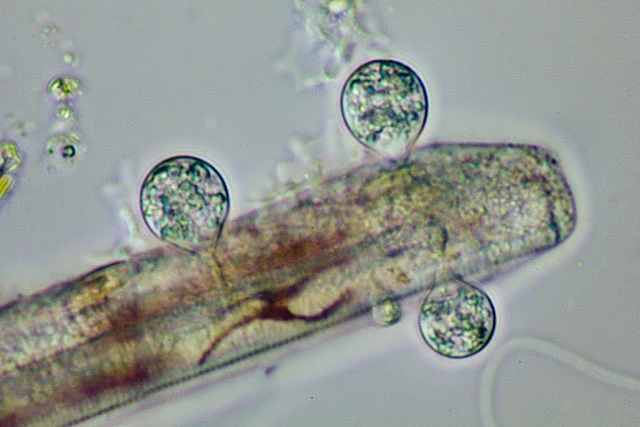
Diagnosis: Trophozoites extremely variable in shape and continuously changing the outline; usually flattened and spreading on surfaces. Small advancing individuals sometimes compact. Intermediate-sized cells can show a fan-like outline, can be branched into several arms, or elongate; sometimes drawn out to considerable length, then appearing as two cell bodies just connected via a thin, tense cytoplasmic stand. Due to adhesion on the substrate, moving organisms produce sticky tails, which stretch and finally are retracted. Size extremely variable, ranging from circa 40 µm to over one millimeter; clear distinction between single trophozoites and plasmodia is not possible. Pseudopodia are predominantly produced at the edges of the cell, often originating from a hyaline and very delicate fringe of cytoplasm; sometimes in tufts. Pseudopodia long and thin, tapering, mostly unbranched; dendritic structures and anastomoses occur. No membranosomes present on the pseudopodia. Pseudopodial accumulation indicates direction of cell movement or cell extension, sometimes occurring on several sites in an individual. Coloration of the central cell body ranges from colorless to orange, depending on food source and contraction of the cell body; cell periphery and pseudopodia colourless. Cytoplasm often contains numerous vacuoles, several are contractile; vacuolation sometimes obscured due to numerous tiny refractive granules, but clearly visible at high magnification and in expanded individuals. At least two different populations of cytoplasmic granules distinguishable: colorless granules, possibly corresponding to the membranosomes of Vampyrella. Orange granules, possibly lipid droplets containing dissolved carotenoids of the prey. Numerous nuclei; very inconspicuous, often not visible in living individuals, but appearing as vesicular structures (about 3.5 µm in diameter) in squeezed dying cells. Cells move very smoothly over surfaces accompanied by the occasional retraction of the sticky posterior ends. When food is scarce trophozoites can transform into the isodiametric morphotype, detach and float in the water column. These compact cells, about 30 µm in diameter, resemble Vampyrella due to their radiating pseudopodia.
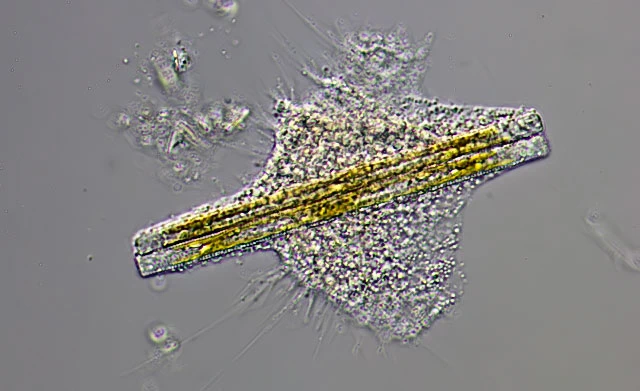
Due to plasmodial organization of unfused trophozoites, difficult to distinguish from the latter; very large plasmodia can exceed one millimeter. Digestive cysts. Cysts vary greatly in size and shape depending of type and amount of prey. The smallest cysts are elliptical or roundish and about 30 mm in size, whereas large cysts can reach several hundred microns in length. Larger cysts can be elongated, irregular lobed, or dumbbell-shaped; when single large algal cells are engulfed, the cyst outline resembles the prey, as shown in case of banana-shaped cysts containing Closterium sp. or very slender cysts containing Synedra sp. The digestive cysts exhibit only one tight fitting cyst envelope, with even surface; corresponding to the inner cyst envelope of Vampyrella. Young stages reveal the green algal prey in the centre of the cell surrounded by colorless or slightly orange cytoplasm, in older stages the ingesta turned brown. During hatching of the trophozoites the food remnants are left behind in the empty cyst envelope. Although internal cell division occurs, the daughter cells occasionally fuse again outside of the cyst envelope during hatching. No resting cysts observed.
Ecology: Fresh water and soil. Observed food organisms: Cylindrocystis brebissonii, Closterium cornu, Closterium sp., Planotaenium interruptum, Saccharomyces cerevisiae, several diatoms and fragments of filamentous algae.
Remarks: Trophozoites engulf whole prey cells during their movement, often resulting in several food items collected in the cytoplasm of an individual. The occasionally observed change in colour of the prey indicates ongoing digestion before entering the immobile cyst stage.
Source: Hess et al, 2012
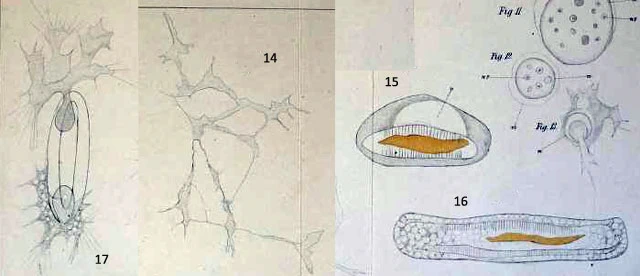
14: reticulum
15-16: Verdauungscysten
17: two individuals leave the cyst.
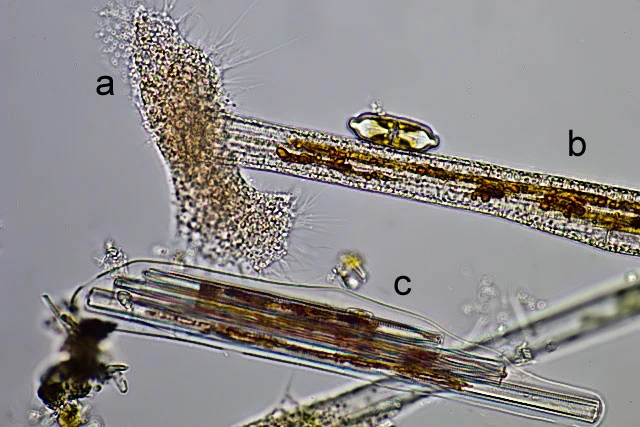
a trophozoit
b cyst
c empty cyst